01Abnormal protein accumulation and aggregate formation are involved in the pathogenesis of various neurodegenerative diseases
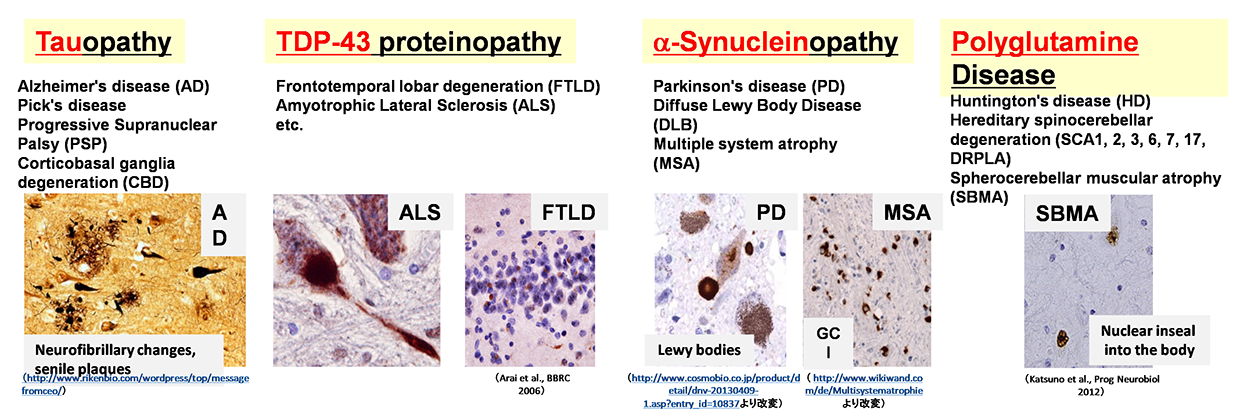
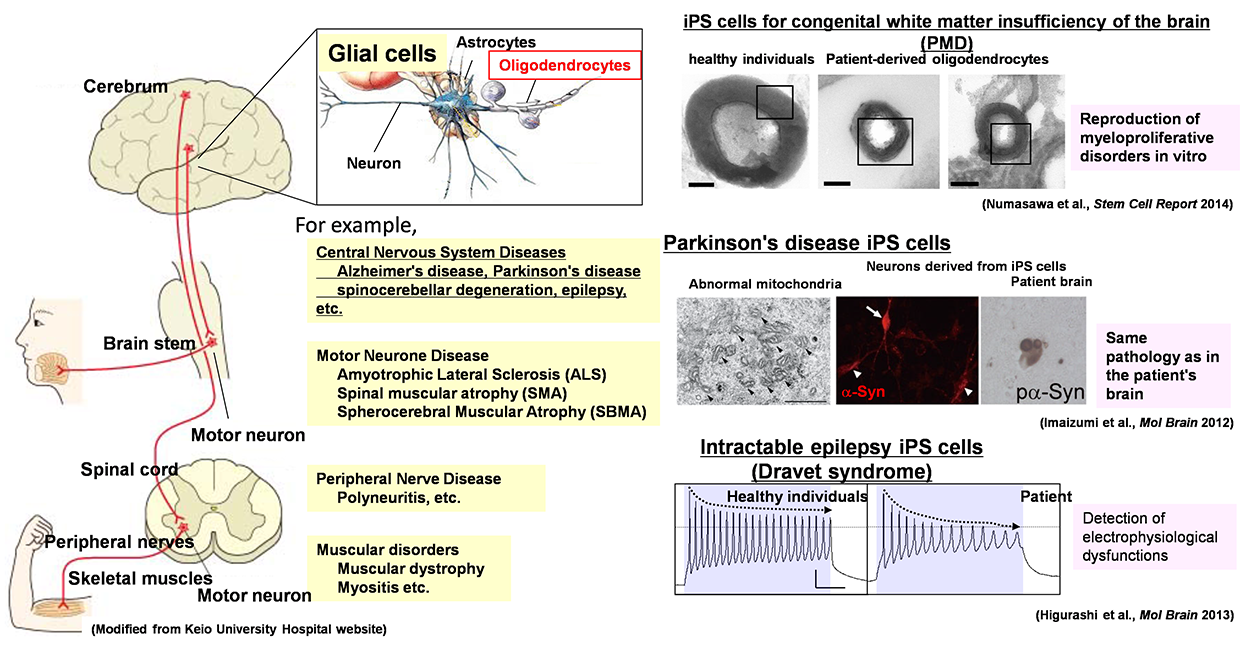
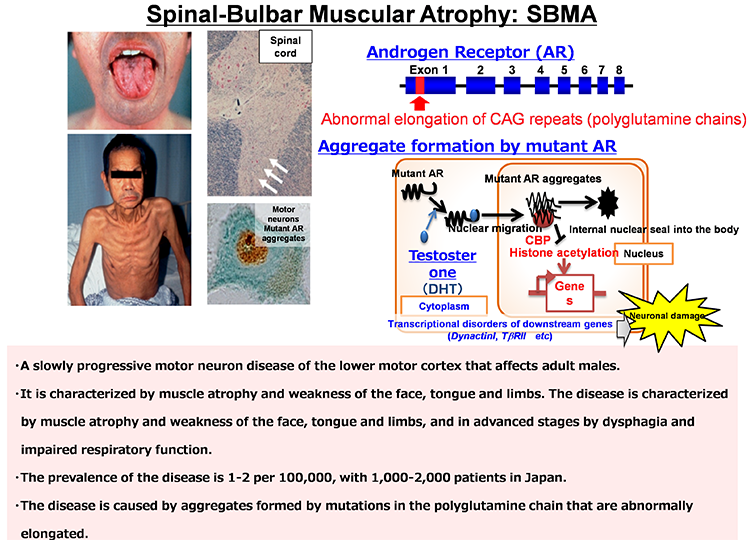
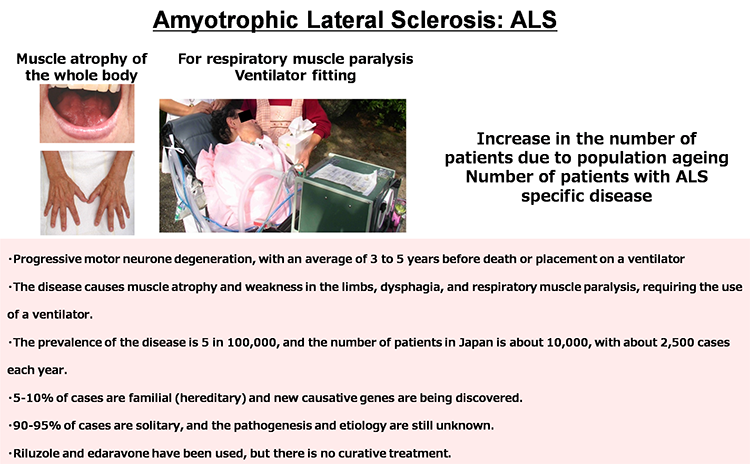
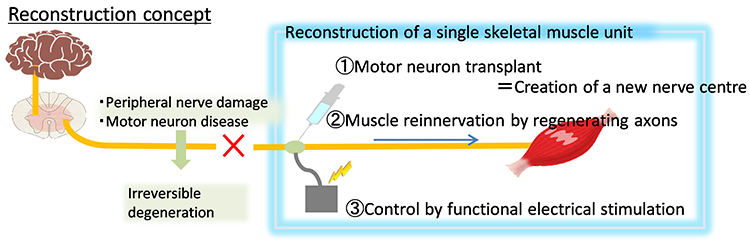
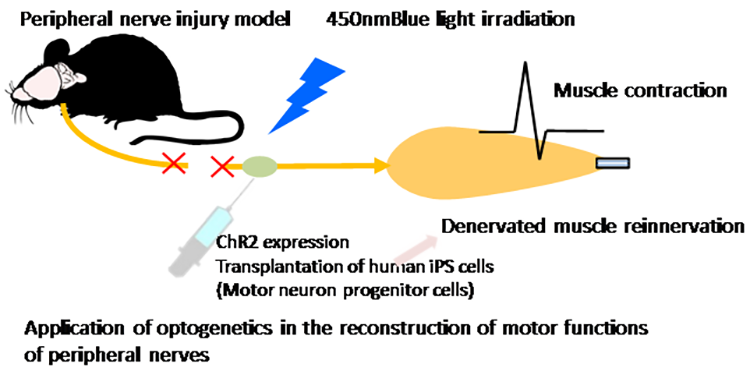
Pathological analyses have shown abnormal protein accumulation in neural tissues of patients with various neurodegenerative diseases. These abnormal proteins are thought to form aggregates upon aging and various stresses, resulting in neurodegeneration. Therefore, many attempts have been made to develop therapeutics targeting the accumulation of these abnormal proteins. However, various findings have suggested that the accumulation of abnormal proteins alone is insufficient to explain the pathogenesis of neurodegeneration. To overcome neurodegenerative diseases, we believe it is necessary to clarify pathogenic events not dependent on the accumulation of abnormal proteins (early-stage pathology). Post-mortem pathology specimens obtained from autopsies are extremely important for investigating the cause of disease but only allow for observations of neurodegeneration after it has occurred; therefore, most human specimens are unsuitable for analyses of disease onset or processes of neurodegeneration. Accordingly, we believe that it is important to use ‘disease models that can reproduce the exact state of the disease’.







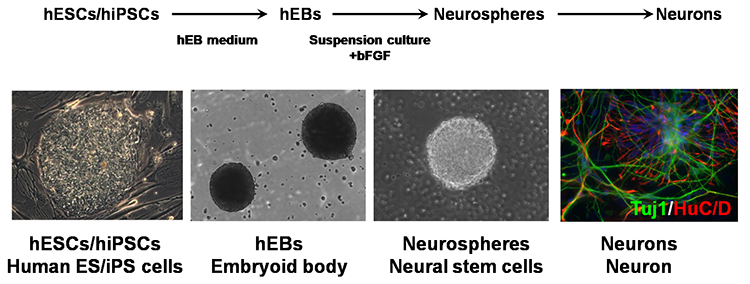
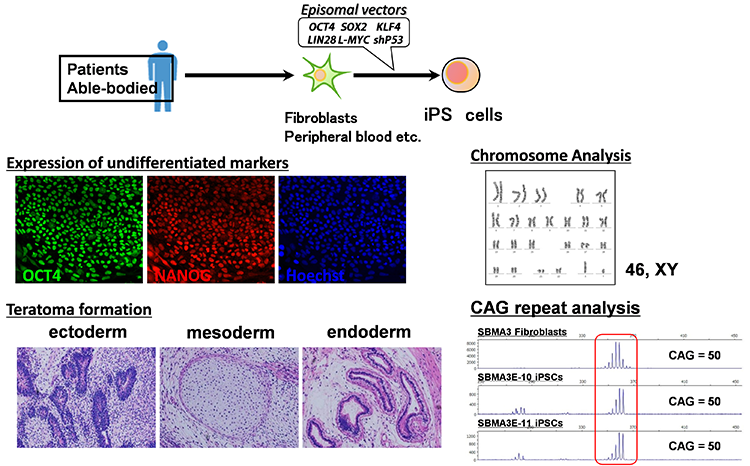
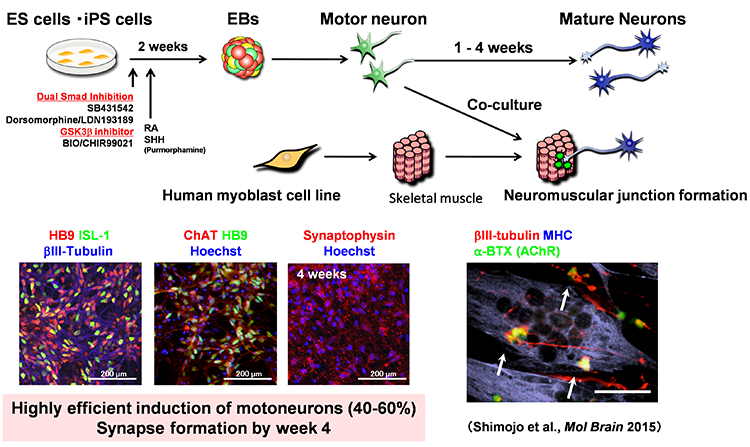
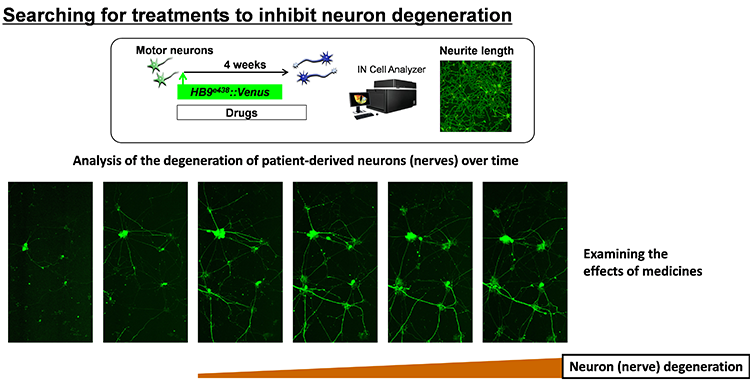
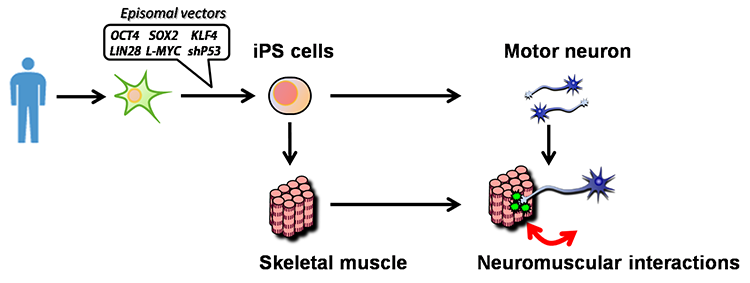
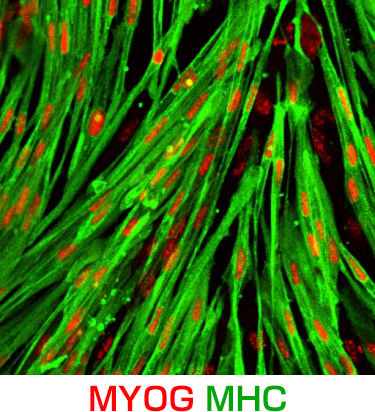
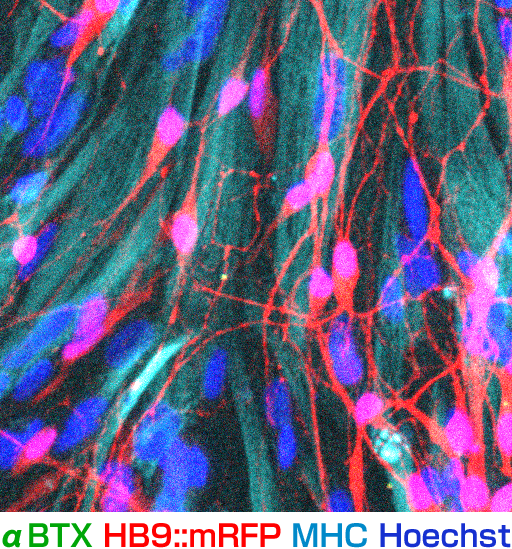
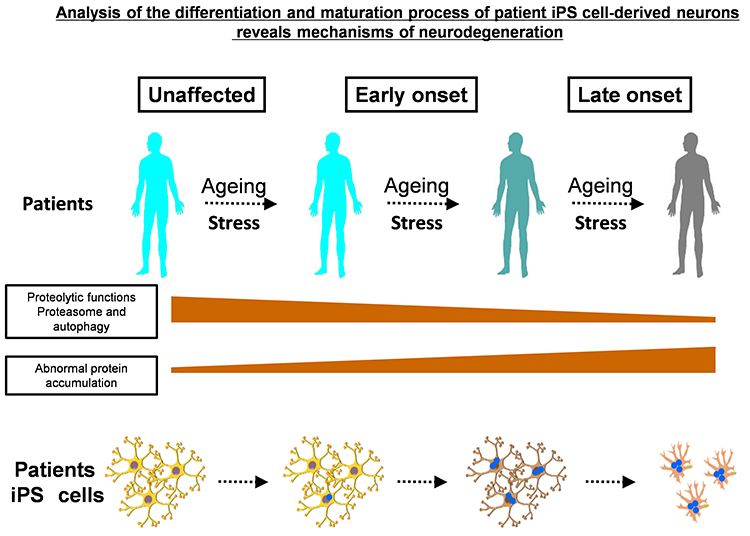 We believe induced pluripotent stem cells (iPSCs) derived from patients with neurological diseases can be turned into neuronal cells as a ‘disease model that can reproduce the exact state of the disease’. To elucidate the pathogenesis of neurological diseases and explore new therapeutic approaches, we are generating iPSCs from somatic cells obtained from patients with neurodegenerative diseases (disease-specific iPSCs) and differentiating ‘patients’ neurons’ from these iPSCs. In particular, we are focusing on processes to differentiate and mature patient-derived iPSCs into neurons that reproduce the onset and progression of diseases (e.g., accumulation or aggregate-formation of abnormal proteins during neuronal degeneration on a culture dish). By employing such techniques, we are creating new disease models that use patient-derived neural cells to elucidate mechanisms of neurodegeneration.
We believe induced pluripotent stem cells (iPSCs) derived from patients with neurological diseases can be turned into neuronal cells as a ‘disease model that can reproduce the exact state of the disease’. To elucidate the pathogenesis of neurological diseases and explore new therapeutic approaches, we are generating iPSCs from somatic cells obtained from patients with neurodegenerative diseases (disease-specific iPSCs) and differentiating ‘patients’ neurons’ from these iPSCs. In particular, we are focusing on processes to differentiate and mature patient-derived iPSCs into neurons that reproduce the onset and progression of diseases (e.g., accumulation or aggregate-formation of abnormal proteins during neuronal degeneration on a culture dish). By employing such techniques, we are creating new disease models that use patient-derived neural cells to elucidate mechanisms of neurodegeneration.